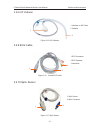
EDAN F6 User manual - 9.2 Maternal Spo
F Series Fetal & Maternal Monitor User Manual Maternal Monitoring
- 89 -
9.2 Maternal SpO
2
Monitoring
9.2.1 Introduction
The monitor provides continuous monitoring of functional arterial oxygen saturation (SpO
2
) and
pulse rate(PR) for pregnant women.
SpO
2
Plethysmogram measurement is employed to determine the oxygen saturation of
hemoglobin in the arterial blood. If, for example, 97% hemoglobin molecules in the red blood
cells of the arterial blood combine with oxygen, then the blood has a SpO
2
oxygen saturation of
97%. The SpO
2
numeric on the monitor will read 97% .The SpO
2
numeric shows the percentage
of hemoglobin molecules which have combined with oxygen molecules to form oxyhemoglobin.
The SpO
2
/PLETH parameter can also provide a pulse rate signal and a plethysmogram wave.
SpO
2
Plethysmogram Measurement Principle:
Pulse oximetry is a continuous and noninvasive monitoring technique used to estimate the
measurement of arterial oxygen saturation. It measures the amount of light penetrating the
patient tissue and reaching the receiver. The reading, obtained through pulse oximetry, uses a
light sensor containing two sources of light (red and infrared) that are absorbed by
hemoglobin and transmitted through tissues to a photodetector.
The amount of light penetrated depends on multiple factors and most of them are constant.
However, the arterial blood flow changes with time passing by as is pulsative. The arterial
oxygen saturation can be obtained through testing the absorbed light during pulsation.
Plethysmogram wave and pulse rate signal can be also provided during pulsation testing.
The sensor contains LEDs that emit red light at a wavelength of approximately 660 nm and
infrared light at a wavelength of approximately 900 nm. Information about wavelength range can
be especially useful to clinicians.
The monitor is compatible with the SpO
2
sensors supplied by the manufacturer only, and the
provided SpO
2
sensor can only be used with this monitor.
Compatibility should be checked prior to use. Otherwise the monitor performance can be
degraded.
They have been tested and found to comply with the limits for medical device in
IEC/EN60601-1-2 (International standard for EMC testing of Medical Electrical Equipment,
second edition). These limits are designed to provide reasonable protection against harmful
interference in typical medical installation.
WARNING
1 Before monitoring, check whether the sensor cable is normal. If any sign of damage in
the SpO
2
sensor is detected, do not use the sensor. Return it to the manufacturer for
service.
2 Do not put the SpO
2
sensor on the extremities with arterial catheter or venous syringe.