EDAN F6 User manual - page 183
F Series Fetal & Maternal Monitor User Manual Ordering Information
- 172 -
Appendix 6 Ordering Information
Accessories (standard and optional configuration) supplied or approved by the manufacturer can
be used with the monitor. See the following table for details. The accessories employed by us,
such as the rechargeable battery, are products having passed the authentication of CE, and they
have the characteristics specified by their manufacturers.
The materials with which the patient
can come into contact conform to the standard of ISO 10993.
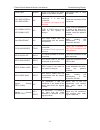
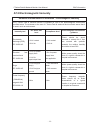
Accessory (Spare Part)
Part Number
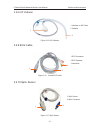
US Transducer 1 (purple label, for F9/F9 Express)
02.01.31528
US Transducer 2 (yellow label, for F9/F9 Express)
02.01.107705
US Transducer (pink label, for F6/F6 Express) 02.01.109301
Wireless US Transducer (American Standard)
02.01.000925
Wireless TOCO Transducer (American Standard)
02.01.000926
FTS-3 Overall Unit
83.62.001974
FTS-3 Overall Unit (Singleton Pregnancy)
83.62.002459
TOCO Transducer
02.01.31527
TOCO Transducer (IUP)
02.01.107791
Remote Event Marker
02.01.210095
Belt
01.57.471447
DECG Cable
01.13.036358
Disposable Fetal Spiral Electrode
01.57.02145
Disposable Attachment Pad Electrode
01.57.02146
IUP Cable
01.13.104152
IUP Connecting Cable
01.13.036357
Disposable Intrauterine Pressure Catheter
01.57.104153
3-lead ECG Cable (Grabber style, IEC)
01.57.471098
3-lead ECG Cable (Grabber style, AHA)
01.57.471095