Abbott i-STAT 1 System Manual - page 429
Rev. Date: 03-Aug-12
Art: 714177-00N
GLUCOSE/GLU
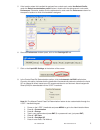
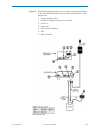
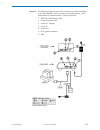
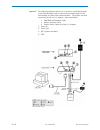
Glucose is measured amperometrically. Oxidation of glucose, catalyzed by the enzyme glucose oxidase,
produces hydrogen peroxide (H
2
O
2
). The liberated hydrogen peroxide is oxidized at the electrode to produce
a current which is proportional to the sample glucose concentration.
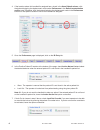
glucose oxidase
β-D-glucose + H
2
O + O
2
D-gluconic acid + H
2
O
2
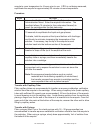
H
2
O
2
2H
+
+ O
2
+ 2e
-
See below for information on factors affecting results. Certain substances, such as drugs, may affect analyte
levels in vivo.
1
If results appear inconsistent with the clinical assessment, the patient sample should be retested using
another cartridge.
Intended Use
The test for glucose, as part of the i-STAT System, is intended for use in the in vitro quantification of glucose
in arterial, venous, or capillary whole blood.
Glucose measurements are used in the diagnosis, monitoring, and treatment of carbohydrate metabolism
disorders including, but not limited to, diabetes mellitus, neonatal hypoglycemia, idiopathic hypoglycemia,
and pancreatic islet cell carcinoma.
Contents
Each i-STAT cartridge contains one reference electrode (when potentiometric sensors are included in the
cartridge configuration), sensors for the measurement of specific analytes, and a buffered aqueous calibrant
solution that contains known concentrations of analytes and preservatives. For cartridges that contain a
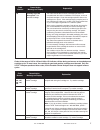
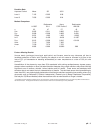
sensor for the measurement of glucose, a list of reactive ingredients is indicated below:
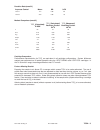
Reactive Ingredient
Biological Source
Glucose
N/A
Glucose Oxidase
Aspergillus niger
Metrological Traceability
The i-STAT System test for glucose measures glucose amount-of-substance concentration in the plasma
fraction of arterial, venous, or capillary whole blood (dimension mmol L
-1
) for in vitro diagnostic use.
Glucose values assigned to i-STAT’s controls and calibration verification materials are traceable to the U.S.
National Institute of Standards and Technology (NIST) standard reference material SRM965. i-STAT System
controls and calibration verification materials are validated for use only with the i-STAT System and assigned
values may not be commutable with other methods. Further information regarding metrological traceability
is available from Abbott Point of Care Inc..