Abbott i-STAT 1 System Manual - page 431
Rev. Date: 03-Aug-12
Art: 714177-00N
Glu - 3
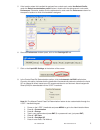
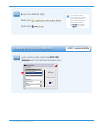
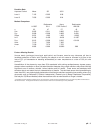
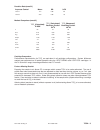
Precision Data (mg/dL)
Aqueous Control
Mean
SD
%CV
Level 1
41.8
0.68
1.6
Level 3
289
2.4
0.8
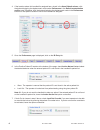
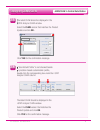
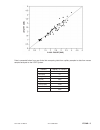
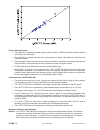
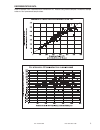
Method Comparison (mg/dL)
Beckman Coulter
LX20
Bayer 860
Dade Dimension
RxL-Xpand
n
35
40
32
Sxx
2.21
4.71
0.98
Syy
0.69
0.96
0.59
Slope
1.03
0.99
1.01
Int’t
-3.39
-1.67
-0.85
Sy.x
0.91
0.70
1.57
Xmin
45
58
48
Xmax
297
167
257
r
0.999
0.993
0.998
Cartridge Comparison
The performance characteristics of the sensors are equivalent in all cartridge configurations. System
difference analysis was performed on 34 patient samples using the i-STAT CHEM8+ and i-STAT CG8+
cartridges. In the 65–249 mg/dL range, the average difference was 0.80.
Factors Affecting Results*
Glucose values will decrease in whole blood samples over time. Venous blood glucose is as much as
7 mg/dL less than capillary blood glucose as a result of tissue utilization.
6
Test concentrations used were as per the CLSI guidance document,
7
unless otherwise indicated.
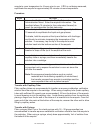
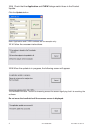
The following substances are known to interfere with the i-STAT glucose assay:
Substance
Test Concentration
(mmol/L)
Interference
Acetaminophen
1.32
Increased i-STAT Glucose results. See
Note Below
Acetylcysteine
10.2
Decreased i-STAT Glucose results. See
Note below.
Bromide
37.5
Decreased i-STAT Glucose results. See
Note below.
Bromide (therapeutic)
2.5
8,9,10
Decreased i-STAT Glucose results by
approximately 5 mg/dL. See Note below.
Hydroxyurea
0.92
Increased i-STAT Glucose results. Use
another method.
Thiocyanate
6.9
Decreased i-STAT Glucose results by
approximately 7 mg/dL.
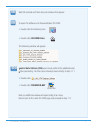
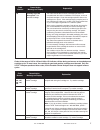
The following substances are known not to significantly interfere with the i-STAT glucose assay at
the stated test concentrations:
Substance
Test Concentration
(mmol/L)
Acetaldehyde
0.045
11
Acetaminophen (therapeutic)
0.132
11
Acetoacetate
2.0
Acetylcysteine (therapeutic)
0.3
12,13
Ascorbate
0.34
Dopamine
0.006
Formaldehyde
0.133
11