Abbott i-STAT 1 System Manual - page 563
3
Art: 715209-00E
Rev. Date: 06/11/08
Because the manufacturing process for the cartridges produces consistent lots with little lot-to-lot
variation, Abbott Point of Care Inc. does not make any recommendations for this procedure. Each
new lot of cartridges should be assessed using the i-STAT quality control solutions according to the
procedure in the System Manual.
troubleShootIng
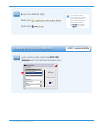
Should quality control sample results fall outside of the acceptable ranges, calibration verification samples
with very low or very high concentrations could be helpful in characterizing a reagent problem. This
information would be most valuable if the user could affect a change in the calibration of the test system to
correct the problem.
The i-STAT System was designed so that the intended users, who are not familiar with laboratory
procedures, cannot make any adjustments that would affect results. The characteristics of the sensors
are well known and results of control solutions would be sufficient for Technical Support specialists to help
users resolve control out-of-range problems.
VerIfyIng that reSultS haVe not been affected by maIntenance or repaIr
procedureS
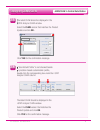
In multi-use systems, reagents and samples are run through the analyzer and, therefore, there should be a
method of checking that all components are performing together according to specifications after any one
component is affected. Calibration verification solutions can be used to verify that the calibration or slope
of the measuring system has not been affected by the maintenance or repair procedure.
The user cannot perform any maintenance procedures on the i-STAT System. However, the software in
the analyzer is updated periodically – a procedure that potentially could cause a change in results. In
fact, software updates are released to ensure that results do not change over time. Calibration verification
solutions could be tested to verify that results have not been affected. This procedures should be
suspended or replaced with controls once it has been determined that software updates do not affect
results. Since i-STAT has been effectively updating software for over 10 years, this procedure is not part of
Abbott Point of Care Inc’s. recommended quality program.
Repaired and newly purchased analyzers are received with factory calibration. Again, the Electronic
Simulator can better assure that the analyzer’s most important function is within factory specifications than
calibration verification or control solutions.
Testing calibration verification samples or comparing patient sample results on a new or repaired analyzer
with an older analyzer will assess cartridge performance only. Any variations in analyzer performance will
not be statistically discernable above the performance of the cartridges. When multiple analyzers are to be
used at a facility, Abbott Point of Care Inc. recommends including at least two analyzers in any performance
verification studies so that statistics reflect the “system.”