Abbott i-STAT 1 System Manual - page 645
Manual is about: For use with a CLIA Certificate of Waiver
of
658
PROCEDURE MANUAL FOR THE i-STAT SYSTEM
24
REV. DATE: 16-Oct-12
ART: 714446-00L
Remedial Action
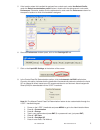
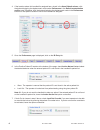
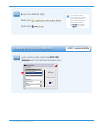
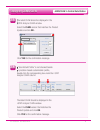
If any results are outside the published expected ranges:
DO NOT USE cartridges from the suspect lot.
Quarantine the suspect lot.
Notify the i-STAT System Coordinator immediately.
Record the QC failure in the i-STAT QC Action Log along with the action taken.
CALIBRATION
For cartridges, calibration is automatically performed as part of the test cycle on each cartridge type, except coagulation and
immunoassay cartridges. Operator intervention is not necessary.