Abbott i-STAT 1 System Manual - page 539
3
Art: 714262-00AA
Rev. Date: 17-Mar-11
SAmPLe hAndLIng
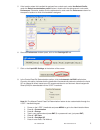
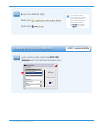
It is especially important not to expose aqueous samples for blood gases and ionized calcium to air. The
instructions in the Quality Control section of the i-STAT System Manual under “Transfer with Capillary Tube”
or “Transfer with Syringe” should be followed.
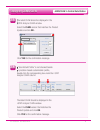
• Take care not to draw air bubbles into the transfer device. If there is only one ampule per level, the
opened ampule may be exposed to air for too long to fill a second transfer device. Exposure to air
could affect pH,
PCO2, PO2 and ionized calcium results.
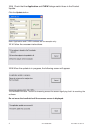
• If air is trapped between the leading edge of the solution and the syringe plunger, do not invert
the syringe to expel it. Air trapped near the plunger will not affect results from the solution near the
tip of the syringe, but may affect the sample as it passes through the syringe when being expelled.
• One or two drops should be expelled from a syringe before filling a cartridge.
PRofICIenCy TeST fAILuReS
Certain PT survey samples are not compatible with i-STAT System sensors and may contain interfering
substances that could affect results. Fluorocarbon samples for blood gases and fixed-cell samples for
hematocrit are not compatible with the sensors.
Occasionally, results from non-whole blood samples may differ between older and newer generations
of cartridges. These differences may be significant. Abbott Point of Care will work with PT providers
to prevent i-STAT System users from being unfairly penalized for PT failures that can be attributed to
manufacturing changes. This is why it is important to record the cartridge type and lot numbers used
to test samples. Note: the CLEW software prevents manufacturing changes from affecting results when
testing patient samples.
PRofICIenCy TeSTIng SuRvey PRovIdeRS
Note 1: This list may not include all providers and is not intended as an endorsement of any particular
provider. Abbott Point of Care no longer evaluates surveys and assumes that for those surveys
that list the i-STAT System, compatibility has been confirmed by third-party testing or a history
of adequate results.
Note 2: Select a survey for hematocrit which is formulated for a conductometric method
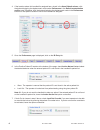
Proficiency Provider
Survey Title(s)
Additional Information or
Recommendations
College of American Pathologists (CAP)
College of American
Pathologists (CAP)
325 Waukegan road
Northfield, IL
60093-2750
800-323-4040 or 847-832-7000
www.cap.org
CAP AQ3 survey for blood gases and
electrolytes: includes iCa, Cl, Hct, Hb,
Lac,
PCO
2
, pH,
PO
2
, K, Na, and TCO
2
CAP AQ4 survey for blood gases and
electrolytes: includes all AQ3 analytes
plus Glu, BUN, and Crea.
CAP Excel L24: includes iCa, Cl, Hct,
PCO
2
, pH,
PO
2
, K, and Na.
AQ3 or AQ4 is recommended if L24
does not cover all analytes
PO2 results should not be
corrected for barometric pressure.
The barometric pressure should
be reported with the uncorrected
results. Barometric pressure can
be reported from the Status Page
after the results are displayed.
Note: Use the green labeled
samples for hematocrit testing.
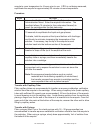
CAP CT1 survey for Activated Clotting
Time (ACT)
Samples are reconstituted with
0.025 mol/L (25 mmol/L ) calcium
chloride
supplied by the user.
CAP Plasma Cardiac Markers (PCArM)
Survey
XL/XLN Excel Series L30
For use with i-STAT cTnI, CK-MB,
and BNP cartridges.
Whole Blood PT/INr (WP3) and Excel
PT/INr XC19