Abbott i-STAT 1 System Manual - page 529
Rev. Date: 23-May-12
Art: 716969-00J
BNP - 11
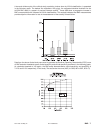
Plasma Blood
Sample
Concentration
pg/mL)
Diluted Concentration
(pg/mL)
% Recovery
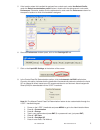
A
590
—
—
B
2764
—
—
C
5123
—
—
A+B
—
1570
94%
B+C
—
3992
101%
A+C
—
2734
96%
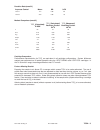
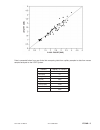
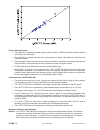
A plasma sample was spiked with BNP to a value of approximately 5000 pg/mL and the concentration was
determined by duplicate measurements with i-STAT BNP test cartridges; the result was found to be within
200 pg/mL of the intended target. This sample was subjected to a series of dilutions with fresh, un-spiked
plasma in order to prepare a range of concentrations. The concentration of each sample/dilution was
calculated based on the measured concentration of the initial solution and the dilutions performed. The
diluted samples were then measured in i-STAT BNP test cartridges (N = 6-10). The procedure was repeated
with a whole blood sample. The results of these experiments are summarized in the following table.
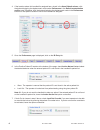
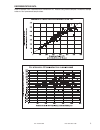
Sample
Dilution
Calculated [BNP]
(pg/mL)
Measured [BNP]
(pg/mL)
%Recovery
Plasma
1
52
57
110%
Plasma
2
104
114
110%
Plasma
3
259
265
103%
Plasma
4
518
560
108%
Plasma
5
1036
1002
97%
Plasma
6
2072
2277
110%
Plasma
7
3107
3384
109%
Plasma
8
4143
4222
102%
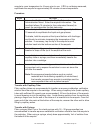
Whole Blood
1
44
41
93%
Whole Blood
2
88
88
100%
Whole Blood
3
269
287
107%
Whole Blood
4
537
554
103%
Whole Blood
5
725
720
99%
Whole Blood
6
1450
1367
94%
Whole Blood
7
3042
2826
93%
Whole Blood
8
4056
3856
95%
Test Limitations
The frequency of suppressed results is affected by atmospheric pressure. Suppressed result rates may
increase with higher elevations (decreased barometric pressure) and may become persistent if testing is
performed at more than 7500 feet above sea level. Where unavailability of results is unacceptable, i-STAT
recommends having an alternate test method available.
Samples from patients who have been exposed to animals or who have received therapeutic or diagnostic
procedures employing immunoglobulins or reagents derived from immunoglobulins may contain antibodies,
e.g. HAMA or other heterophile antibodies, which may interfere with immunoassays and produce erroneous
results.
28-34
The generation of potentially interfering antibodies in response to bacterial infections has been
reported.
28
While this product contains reagents that minimize the effect of these interferents, and QC
algorithms designed to detect their effects, the possibility of interference causing erroneous results should
be evaluated carefully in cases where there are inconsistencies in the clinical information.