Abbott i-STAT 1 System Manual - page 473
Rev. Date: 03-Aug-12
Art: 714184-00M
LAC - 3
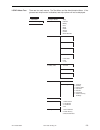
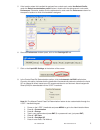
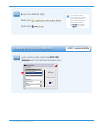
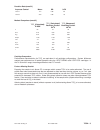
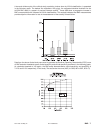
Precision Data
Aqueous Control
n
Mean
SD
%CV
(mmol/L)
Level
1
120 6.35 0.08 1.21
Level
3
120 0.81 0.03 3.27
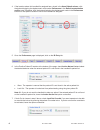
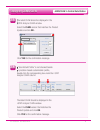
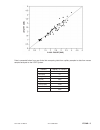
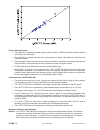
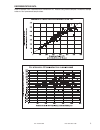
Method Comparison
Radiometer ABL 725
Hitachi 917
(mmol/L)
(whole blood vs.
(i-STAT whole blood vs.
whole blood)
Hitachi plasma)
n
47
47
Sxx
0.123
0.084
Syy
0.136
0.079
Slope
1.02
1.06
Int't
0.12
-0.32
Sy.x
0.18
0.17
Xmin
0.80
1.77
Xmax
14.20
14.24
r
0.998
0.997
Factors Affecting Results*
Special collection procedures are necessary to prevent changes in lactate both during and after the blood
is drawn. For steady state lactate concentrations, patients should be at rest for 2 hours and fasting. Venous
samples should be obtained without the use of a tourniquet or immediately after the tourniquet is applied.
Both venous and arterial samples may be collected into heparinized syringes.
Samples for lactate should be analyzed immediately on drawing as lactate increases by as much as 70%
within 30 minutes at 25 ºC as a result of glycolysis.
2
Test concentrations used were as per the CLSI guidance document,
16
unless otherwise indicated.
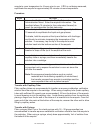
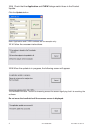
The following substances are known to interfere with the i-STAT lactate assay:
Substance
Test Concentration
(mmol/L)
Interference
Bromide
37.5
Decreased i-STAT lactate results. See
Note below
Glycolic Acid
10.0
17
Increased i-STAT lactate results. Use
another method.
Hydroxyurea
0.92
Increased i-STAT lactate results. Use
another method.
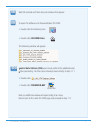
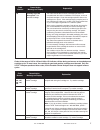
The following substances are known not to significantly interfere with the i-STAT lactate assay at
the stated test concentrations:
Substance
Test Concentration
(mmol/L)
Acetaldehyde
0.045
17
Acetaminophen
1.32
Acetylcysteine
10.2
Ascorbate
0.34
Bromide (therapeutic)
2.5
18-20
Dopamine
0.006
Formaldehyde
0.133
17
β-Hydroxybuterate
6.0
21
Pyruvate
0.31
Salicylate
4.34
Uric Acid
1.4