Abbott i-STAT 1 System Manual - page 447
Rev. Date: 01-Aug-11
Art: 714180-00K
PO
2
AND CALCULATED OXYGEN
SATURATED/sO
2
PO
2
is measured amperometrically. The oxygen sensor is similar to a conventional Clark electrode. Oxygen
permeates through a gas permeable membrane from the blood sample into an internal electrolyte solution
where it is reduced at the cathode. The oxygen reduction current is proportional to the dissolved oxygen
concentration.
See below for information on factors affecting results. Certain substances, such as drugs, may affect analyte
levels in vivo.
1
If results appear inconsistent with the clinical assessment, the patient sample should be retested using
another cartridge.
Intended Use
The test for
PO
2
, as part of the i-STAT System, is intended for use in the in vitro quantification of oxygen
partial pressure in arterial, venous, or capillary whole blood.
PO
2
measurements are used in the diagnosis, monitoring, and treatment of respiratory disturbances and
metabolic and respiratory-based acid-base disturbances.
Contents
Each i-STAT cartridge contains one reference electrode (when potentiometric sensors are included in the
cartridge configuration), sensors for the measurement of specific analytes, and a buffered aqueous calibrant
solution that contains known concentrations of analytes and preservatives.
Metrological Traceability
The i-STAT System test for oxygen partial pressure measures oxygen partial pressure in arterial, venous, or
capillary whole blood (dimension kPa) for in vitro diagnostic use.
PO
2
values assigned to i-STAT’s controls
and calibration verification materials are traceable to U.S. National Institute of Standards and Technology
(NIST) standard reference materials via commercially available certified specialty medical gas standards.
i-STAT System controls and calibration verification materials are validated for use only with the i-STAT
System and assigned values may not be commutable with other methods. Further information regarding
metrological traceability is available from Abbott Point of Care Inc..
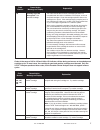
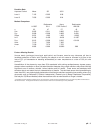
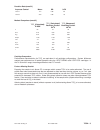
Expected Values
Reportable Reference
Test
Units* Range Range
2
PO
2
mmHg
5 – 800
80 – 105
kPa
0.7 – 106.6
10.7 – 14.0
sO
2
**
%
not applicable
95 – 98
*The i-STAT System can be configured with the preferred units.
** Calculated
To convert
PO
2
results from mmHg to kPa, multiply the mmHg value by 0.133.