Abbott i-STAT 1 System Manual - page 487
Rev. Date: 31-Jan-12
Art: 715878-00M
Kaolin ACT - 3
Method comparison data were collected using a modification of the CLSI guideline EP9-A
2
. Venous or
arterial blood samples were collected in plastic syringes and analyzed in duplicate on the i-STAT System
and in duplicate using the comparative methods. All samples were analyzed immediately upon collection.
The patient populations in the studies were those in which ACT is routinely used and included both aprotinin
and non-aprotinin receiving patients. All were undergoing cardiac surgery. Sample types included baseline,
heparin-treated, and heparin-reversed samples.
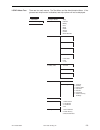
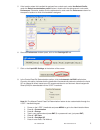
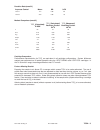
Deming regression analysis
3
was performed on the first replicate of each sample. In the method comparison
table, n is the number of specimens in the data set, Sxx and Syy refer to estimates of the imprecision based
on the duplicates of the comparative and i-STAT methods respectively, Sy.x is the standard error of the
estimate, and r is the correlation coefficient.
Method comparisons will vary from site to site due to differences in the sample handling, reagent and
instrument systems in use, and other site-specific variables.
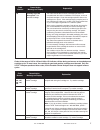
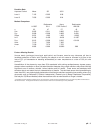
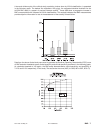
Hemochron FTK-ACT
CVOR
Site 1
Site 2
Site 3
n
104
118
106
Sxx
9.1%
6.8%
7.6%
Syy
3.6%
4.0%
3.6%
Slope
0.96
1.05
0.96
Intercept
-12
-38
-39
Xmin
68
111
81
Xmax
1286
1310
1102
r
0.906
0.940
0.971
Factors Affecting Results*
The i-STAT
Kaolin
ACT test is not significantly prolonged in the presence of aprotinin (Trasylol).
*It is possible that other interfering substances may be encountered. These results are representative and your results may differ somewhat due to
test-to-test variation. The degree of interference at concentrations other than those listed might not be predictable.
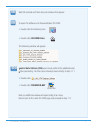
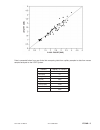
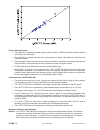
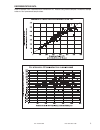
Heparin sensitivity was demonstrated using whole blood samples to which varying concentrations of
heparin were added in vitro.
The following three graphs below each indicate the response of a different donor with respect to heparin
concentration: